Comunidades bacterianas del intestino de escarabajos peloteros y del suelo en sucesión en la Reserva Tapichalaca
DOI:
https://doi.org/10.54753/cedamaz.v13i1.1795Resumen
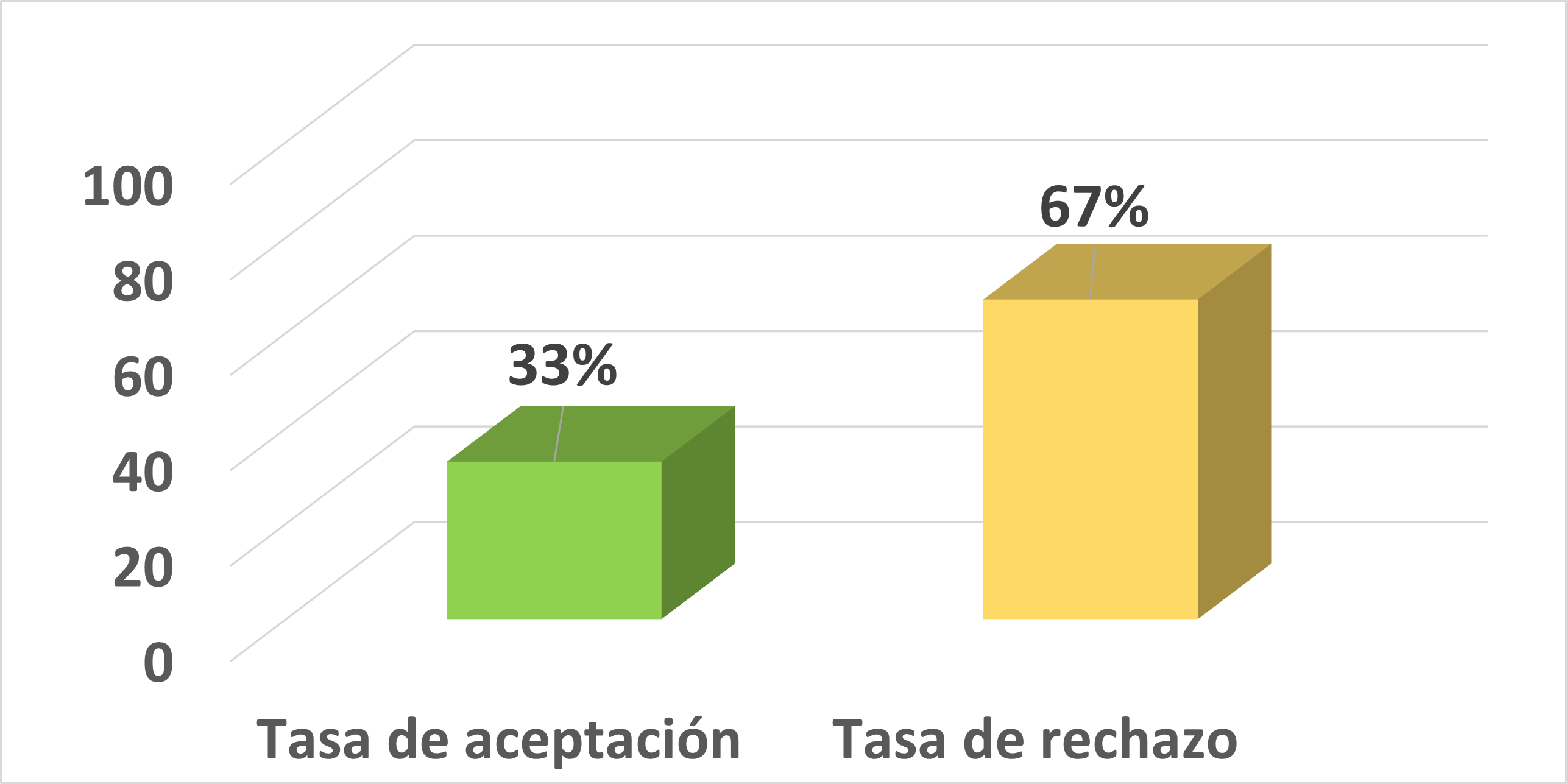
El suelo es un ecosistema importante en el que diferentes especies bacterianas desempeñan un papel fundamental como bioindicadores de su comportamiento ecológico y evolutivo. En el presente estudio se aislaron y caracterizaron morfológicamente comunidades bacterianas de la microbiota intestinal de escarabajos peloteros (Coleoptera: Scarabainae) y del suelo de tres estados de sucesión ecológica (bosque primario, bosque restaurado y pastizal en restauración) de la Reserva Natural Tapichalaca. Se llevó a cabo el aislamiento bacteriano de muestras de suelo y de intestino de escarabajos mediante la inoculación de cultivos mixtos y puros en dos medios de cultivo: Agar Nutritivo (AN) y Agar Tripticasa de Soya (TSA). La identificación morfológica de las colonias bacterianas se realizó a través de la observación de características macroscópicas y la identificación de las células bacterianas mediante Tinción Gram y observación microscópica. Las cepas bacterianas obtenidas en el aislamiento de consorcios bacterianos de las muestras del intestino de los escarabajos y de las muestras de suelo, se asociaron a diferentes posibles géneros bacterianos y se logró concluir que existe variación entre algunos de los géneros identificados en relación con los tres estados de sucesión ecológica. En las muestras intestinales de escarabajos, de los cinco posibles géneros asignados, el género Clostridium se identificó en el bosque primario y Enterobacter en la muestra intestinal de un espécimen del pastizal. En las muestras de suelo, el género Corynebacterium se asoció en la muestra de suelo del bosque primario y Pseudomonas en la muestra de suelo del pastizal.Citas
Arias-Cordero, E., Ping, L., Reichwald, K., Delb, H., Platzer, M., & Boland, W. (2012). Comparative Evaluation of the Gut Microbiota Associated with the Below- and Above-Ground Life Stages (Larvae and Beetles) of the Forest Cockchafer, Melolontha hippocastani. PLoS ONE, 7(12). https://doi.org/10.1371/journal.pone.0051557
Bollinger, A., Thies, S., Katzke, N., & Jaeger, K. (2020). The biotechnological potential of marine bacteria in the novel lineage of Pseudomonas pertucinogena. Microbial Biotechnology, 13(1), 19–31. https://doi.org/10.1111/1751-7915.13288
Briones-Roblero, C. I., Hernández-García, J. A., Gonzalez-Escobedo, R., Soto-Robles, L. V., Rivera-Orduña, F. N., & Zúñiga, G. (2017). Structure and dynamics of the gut bacterial microbiota of the bark beetle, Dendroctonus rhizophagus (Curculionidae: Scolytinae) across their life stages. PLOS ONE, 12(4), e0175470. https://doi.org/10.1371/journal.pone.0175470
Castañeda Briones, M. T. (2003). (PDF) Microbiología Aplicada: Manual de Laboratorio, Castañeda Briones, María Teresa - DOKUMEN.TIPS. https://dokumen.tips/education/microbiologia-aplicada-manual-de-laboratorio-castaneda-briones-maria-teresa.html?page=1
Chamorro, W., Marín-Armijos, D., Granda, V., & Vaz-De-Mello, F. Z. (2018). Checklist with a key to genera and subgenera of dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae) present and supposed for Ecuador. Revista Colombiana de Entomología, 44(1), 72–100. https://doi.org/10.25100/SOCOLEN.V44I1.6545
Chouaia, B., Goda, N., Mazza, G., Alali, S., Florian, F., Gionechetti, F., Callegari, M., Gonella, E., Magoga, G., Fusi, M., Crotti, E., Daffonchio, D., Alma, A., Paoli, F., Roversi, P. F., Marianelli, L., & Montagna, M. (2019). Developmental stages and gut microenvironments influence gut microbiota dynamics in the invasive beetle Popillia japonica Newman (Coleoptera: Scarabaeidae). Environmental Microbiology, 21(11), 4343–4359. https://doi.org/10.1111/1462-2920.14797
Chu, H., Fujii, T., Morimoto, S., Lin, X., Yagi, K., Hu, J., & Zhang, J. (2007). Community structure of ammonia-oxidizing bacteria under long-term application of mineral fertilizer and organic manure in a sandy loam soil. Applied and Environmental Microbiology, 73(2). https://doi.org/10.1128/AEM.01536-06
Da Silva, P. G., & Hernández, M. I. M. (2015). Spatial patterns of movement of dung beetle species in a tropical forest suggest a new trap spacing for dung beetle biodiversity studies. PLoS ONE, 10(5), 1–18. https://doi.org/10.1371/journal.pone.0126112
Delgado-Baquerizo, M., Maestre, F. T., Reich, P. B., Jeffries, T. C., Gaitan, J. J., Encinar, D., Berdugo, M., Campbell, C. D., & Singh, B. K. (2016). Microbial diversity drives multifunctionality in terrestrial ecosystems. Nature Communications, 7. https://doi.org/10.1038/ncomms10541
Ding, G. C., Radl, V., Schloter-Hai, B., Jechalke, S., Heuer, H., Smalla, K., & Schloter, M. (2014). Dynamics of soil bacterial communities in response to repeated application of manure containing sulfadiazine. PLoS ONE, 9(3). https://doi.org/10.1371/journal.pone.0092958
Dong, X., Lv, L., Wang, W., Liu, Y., Yin, C., Xu, Q., Yan, H., Fu, J., & Liu, X. (2019). Differences in distribution of potassium-solubilizing bacteria in forest and plantation soils in myanmar. International Journal of Environmental Research and Public Health, 16(5), 1–14. https://doi.org/10.3390/ijerph16050700
Eaton, W. D., McGee, K. M., Hoke, E., Lemenze, A., & Hajibabaei, M. (2020). Influence of Two Important Leguminous Trees on Their Soil Microbiomes and Nitrogen Cycle Activities in a Primary and Recovering Secondary Forest in the Northern Zone of Costa Rica. Soil Systems, 4(4), 65. https://doi.org/10.3390/soilsystems4040065
Ebert, K. M., Arnold, W. G., Ebert, P. R., & Merritt, D. J. (2021). Hindgut Microbiota Reflects Different Digestive Strategies in Dung Beetles (Coleoptera: Scarabaeidae: Scarabaeinae). Applied and Environmental Microbiology, 87(5), 1–17. https://doi.org/10.1128/AEM.02100-20/SUPPL_FILE/AEM.02100-20-S0001.PDF
Estes, A. M., Hearn, D. J., Snell-Rood, E. C., Feindler, M., Feeser, K., Abebe, T., Dunning Hotopp, J. C., & Moczek, A. P. (2013). Brood Ball-Mediated Transmission of Microbiome Members in the Dung Beetle, Onthophagus taurus (Coleoptera: Scarabaeidae). PLoS ONE, 8(11), e79061. https://doi.org/10.1371/journal.pone.0079061
Falqueto, S. A., de Sousa, J. R., da Silva, R. C., da Silva, G. F., Pinheiro, D. G., & Soares, M. A. (2022). Larval gut microbiome of Pelidnota luridipes (Coleoptera: Scarabaeidae): high bacterial diversity, different metabolic profiles on gut chambers and species with probiotic potential. World Journal of Microbiology and Biotechnology, 38(11). https://doi.org/10.1007/s11274-022-03387-1
Fundación Jocotoco. (2022). Sobre la Reserva Tapichalaca. https://www.jocotoco.org.ec/wb#/ES/Tapichalaca
Garrity, G. M., & Holt, J. G. (2001). The Road Map to the Manual. In Bergey’s Manual® of Systematic Bacteriology. https://doi.org/10.1007/978-0-387-21609-6_15
Gu, Y., Bai, Y., Xiang, Q., Yu, X., Zhao, K., Zhang, X., Li, C., Liu, S., & Chen, Q. (2018). Degradation shaped bacterial and archaeal communities with predictable taxa and their association patterns in Zoige wetland at Tibet plateau. Scientific Reports, 8(1), 3884. https://doi.org/10.1038/s41598-018-21874-0
Guo, Y., Chen, X., Wu, Y., Zhang, L., Cheng, J., Wei, G., & Lin, Y. (2018). Natural revegetation of a semiarid habitat alters taxonomic and functional diversity of soil microbial communities. Science of The Total Environment, 635, 598–606. https://doi.org/10.1016/j.scitotenv.2018.04.171
Heylen, K., Hoefman, S., Vekeman, B., Peiren, J., & De Vos, P. (2012). Safeguarding bacterial resources promotes biotechnological innovation. In Applied Microbiology and Biotechnology (Vol. 94, Issue 3). https://doi.org/10.1007/s00253-011-3797-y
Hu, H., Ye, R., Pang, L., Jiang, H., Tian, K., Gao, Y., Ji, Y., Wan, P., Yang, J., Zou, X., & Tian, X. (2021). After Life Effect of Endophytic Bacterium Bacillus Cereus on Litter Decomposition. Research Square. https://doi.org/10.21203/rs.3.rs-866340/v1
Huang, S., Sheng, P., & Zhang, H. (2012). Isolation and Identification of Cellulolytic Bacteria from the Gut of Holotrichia parallela Larvae (Coleoptera: Scarabaeidae). International Journal of Molecular Sciences, 13(3), 2563–2577. https://doi.org/10.3390/ijms13032563
Kaur, R., & Kaur, S. (2020). Variation in the Phosphate Solubilizing Bacteria from Virgin and the Agricultural Soils of Punjab. Current Microbiology, 77(9), 2118–2127. https://doi.org/10.1007/s00284-020-02080-6
Kudo, R., Masuya, H., Endoh, R., Kikuchi, T., & Ikeda, H. (2019). Gut bacterial and fungal communities in ground-dwelling beetles are associated with host food habit and habitat. The ISME Journal, 13(3), 676–685. https://doi.org/10.1038/s41396-018-0298-3
Larina, G. E., Seraya, L. G., Ivanova, I. O., Poddymkina, L. M., & Vershinin, V. V. (2020). Microbial complex adaptation in soils of different cultivation degrees. IOP Conference Series: Earth and Environmental Science, 579(1). https://doi.org/10.1088/1755-1315/579/1/012068
Li, J., Yu, H., Wu, X., Shen, L., Liu, Y., Qiu, G., Zeng, W., & Yu, R. (2018). Novel Hyper Antimony-Oxidizing Bacteria Isolated from Contaminated Mine Soils in China. Geomicrobiology Journal, 35(8), 713–720. https://doi.org/10.1080/01490451.2018.1454556
Losey, J. E., & Vaughan, M. (2006). The economic value of ecological services provided by insects. In BioScience (Vol. 56, Issue 4). https://doi.org/10.1641/0006-3568(2006)56[311:TEVOES]2.0.CO;2
Nwinyi, O. C., & Akinmulewo, B. A. (2019). Remediation of soil polluted with spent oil using cow dung. IOP Conference Series: Earth and Environmental Science, 331(1), 012058. https://doi.org/10.1088/1755-1315/331/1/012058
Ortiz-Álvarez, R., Fierer, N., de los Ríos, A., Casamayor, E. O., & Barberán, A. (2018). Consistent changes in the taxonomic structure and functional attributes of bacterial communities during primary succession. The ISME Journal, 12(7), 1658–1667. https://doi.org/10.1038/s41396-018-0076-2
Ozoude, T., Eleanya, E., Uzoaru, N., & Okey-Ndeche, N. (2018). Isolation and Characterization of Some Hydrocarbon Utilizing Bacteria Isolated from Contaminated Soil in Zuma, Bwari Area Council, Fct, Abuja, Nigeria. Microbiology Research Journal International, 22(6), 1–8. https://doi.org/10.9734/mrji/2017/34158
Reth, S., Reichstein, M., & Falge, E. (2005). The effect of soil water content, soil temperature, soil pH-value and the root mass on soil CO2 efflux - A modified model. Plant and Soil, 268(1). https://doi.org/10.1007/s11104-005-0175-5
Reynoso, M., Magnoli, C., Barros, G., & Demo, M. (2015). Manual de microbiología general (Primera edición). UniRío. http://www.unirioeditora.com.ar/wp-content/uploads/2018/10/978-987-688-124-1.pdf
Rhoads, D. D., Wolcott, R. D., Sun, Y., & Dowd, S. E. (2012). Comparison of culture and molecular identification of bacteria in chronic wounds. International Journal of Molecular Sciences, 13(3). https://doi.org/10.3390/ijms13032535
Salazar, J. D. G., Quiñones, R., & Jiménez, P. (2009). Aislamiento de Microorganismos del Tracto Digestivo de Larvas de Coleópteros y Lepidópteros Detritívoros y Evaluación, In Vitro, de su Efecto Antagónico en una Cepa de Fusarium oxysporum. Revista Facultad de Ciencias Básicas, 5(1–2), 106–113. https://doi.org/10.18359/RFCB.2124
Sansupa, C., Purahong, W., Wubet, T., Tiansawat, P., Pathom-Aree, W., Teaumroong, N., Chantawannakul, P., Buscot, F., Elliott, S., & Disayathanoowat, T. (2021). Soil bacterial communities and their associated functions for forest restoration on a limestone mine in northern Thailand. PLOS ONE, 16(4), e0248806. https://doi.org/10.1371/journal.pone.0248806
Schloss, P. D., Delalibera, I., Handelsman, J., & Raffa, K. F. (2006). Bacteria associated with the guts of two wood-boring beetles: Anoplophora glabripennis and Saperda vestita (Cerambycidae). Environmental Entomology, 35(3). https://doi.org/10.1603/0046-225X-35.3.625
Shelomi, M., & Chen, M.-J. (2020). Culturing-Enriched Metabarcoding Analysis of the Oryctes rhinoceros Gut Microbiome. Insects, 11(11), 782. https://doi.org/10.3390/insects11110782
Shin, Y., Paek, J., Son, A. Y., Kim, H., Kook, J.-K., Paek, W. K., & Chang, Y. H. (2018). Clostridium composti sp. nov., a new anaerobic bacteria isolated from compost. International Journal of Systematic and Evolutionary Microbiology, 68(12), 3869–3873. https://doi.org/10.1099/ijsem.0.003074
Shukla, S. P., Plata, C., Reichelt, M., Steiger, S., Heckel, D. G., Kaltenpoth, M., Vilcinskas, A., & Vogel, H. (2018). Microbiome-assisted carrion preservation aids larval development in a burying beetle. Proceedings of the National Academy of Sciences of the United States of America, 115(44), 11274–11279. https://doi.org/10.1073/pnas.1812808115
Vasanthakumar, A., Handelsman, J. O., Schloss, P. D., Bauer, L. S., & Raffa, K. F. (2008). Gut microbiota of an invasive Subcortical Beetle, Agrilus planipennis fairmaire, across various life stages. Environmental Entomology, 37(5). https://doi.org/10.1603/0046-225X(2008)37[1344:GMOAIS]2.0.CO;2
Weller, D. M., Raaijmakers, J. M., McSpadden Gardener, B. B., & Thomashow, L. S. (2002). Microbial populations responsible for specific soil suppressiveness to plant pathogens. In Annual Review of Phytopathology (Vol. 40). https://doi.org/10.1146/annurev.phyto.40.030402.110010
Xu, A., Liu, J., Guo, Z., Wang, C., Pan, K., Zhang, F., & Pan, X. (2021). Soil microbial community composition but not diversity is affected by land-use types in the agro-pastoral ecotone undergoing frequent conversions between cropland and grassland. Geoderma, 401(April), 115165. https://doi.org/10.1016/j.geoderma.2021.115165
Yu, Q., Hanif, A., Rao, X., He, J., Sun, D., Liu, S., He, D., & Shen, W. (2021). Long‐term restoration altered edaphic properties and soil microbial communities in forests: evidence from four plantations of southern China. Restoration Ecology, 29(4), 1–11. https://doi.org/10.1111/rec.13354
Zheng, X., Liu, G., Wang, Z., Wang, J., Zhang, H., & Liu, B. (2020). Bacillus dafuensis sp. Nov., Isolated from a Forest Soil in China. Current Microbiology, 77(9), 2049–2055. https://doi.org/10.1007/s00284-020-02014-2
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2023 CEDAMAZ

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-SinDerivadas 4.0.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan los términos siguientes:
-
Luego que el artículo científico es aceptado, para su publicación el o la autora aceptan ceder los derechos de la primera publicación a la Revista CEDAMAZ, conservando sus derechos de autor. Se permite la reproducción total o parcial de los textos que se publican siempre y cuando sea sin fines de lucro. Cuando se ejecute la reproducción total o parcial de los artículos científicos aceptados y publicados en la revista CEDAMAZ, se debe citar la fuente completa y la dirección electrónica de la publicación.
-
Los artículos científicos aceptados y publicados en la revista CEDAMAZ pueden ser depositados por los autores de manera integra en cualquier repositorio sin fines comerciales.
-
Los autores no deben distribuir los artículos científicos aceptados, pero que todavía no han sido publicados oficialmente por la revista CEDAMAZ. En el caso de incumplir esta norma implica el rechazo del articulo científico.
- La publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons